Bent Bond/Antiperiplanar Hypothesis: Modulating the Reactivity and the Selectivity in the Glycosylation of Bicyclic Pyranoside Models
Glycosylation reactions were performed on a series of bicyclic C2-substituted pyranoside models to isolate and analyze factors that control the glycosylation stereoselectivities observed in carbohydrates. The bent bond / antiperiplanar hypothesis (BBAH) orbital model rationalizes all these results by considering hyperconjugation interactions between groups at C2 and the two τ bonds (bent bonds) of oxocarbenium ion intermediates formed under the glycosylation conditions. According to the BBAH, nucleophiles add to the oxocarbenium intermediates by SN2-like antiperiplanar displacement of the weaker of its two τ bonds.
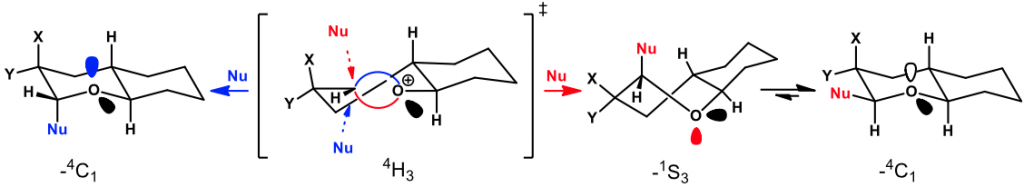
BBAH #10: J.-F. Parent, G. Deslongchamps, P. Deslongchamps, Bent Bond/Antiperiplanar Hypothesis: Modulating the Reactivity and the Selectivity in the Glycosylation of Bicyclic Pyranoside Models. J. Org. Chem., 2020, 85, 4220-4236.
https://doi.org/10.1021/acs.joc.9b03412
Be the first to comment